What Is The Charge On The Ion Formed By Aluminum
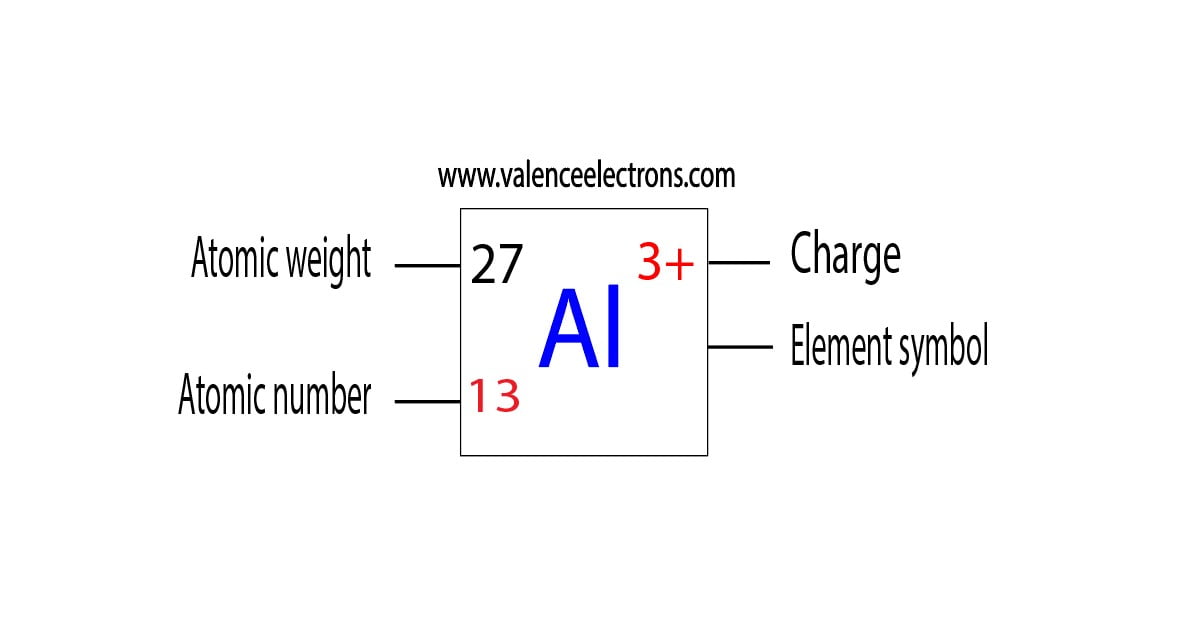
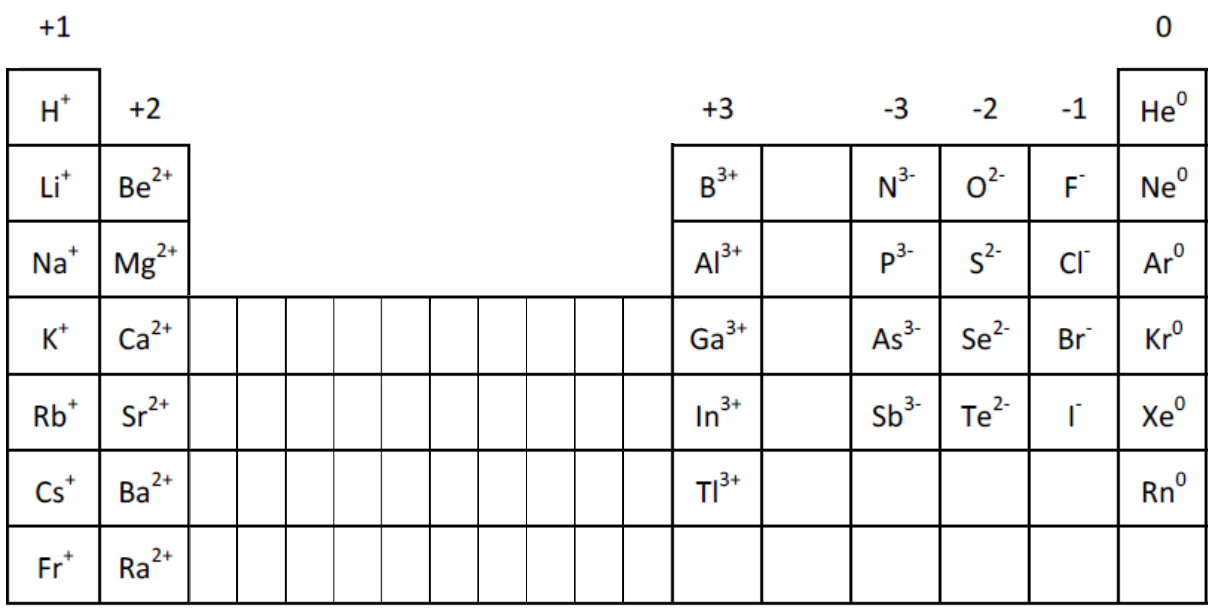
What Is The Charge On The Ion Formed By Aluminum - When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). The charge of an aluminum ion is determined by its electron.
Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron.
When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
What is the Charge on the Ion Formed by Aluminum? Aluminum Profile Blog
When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
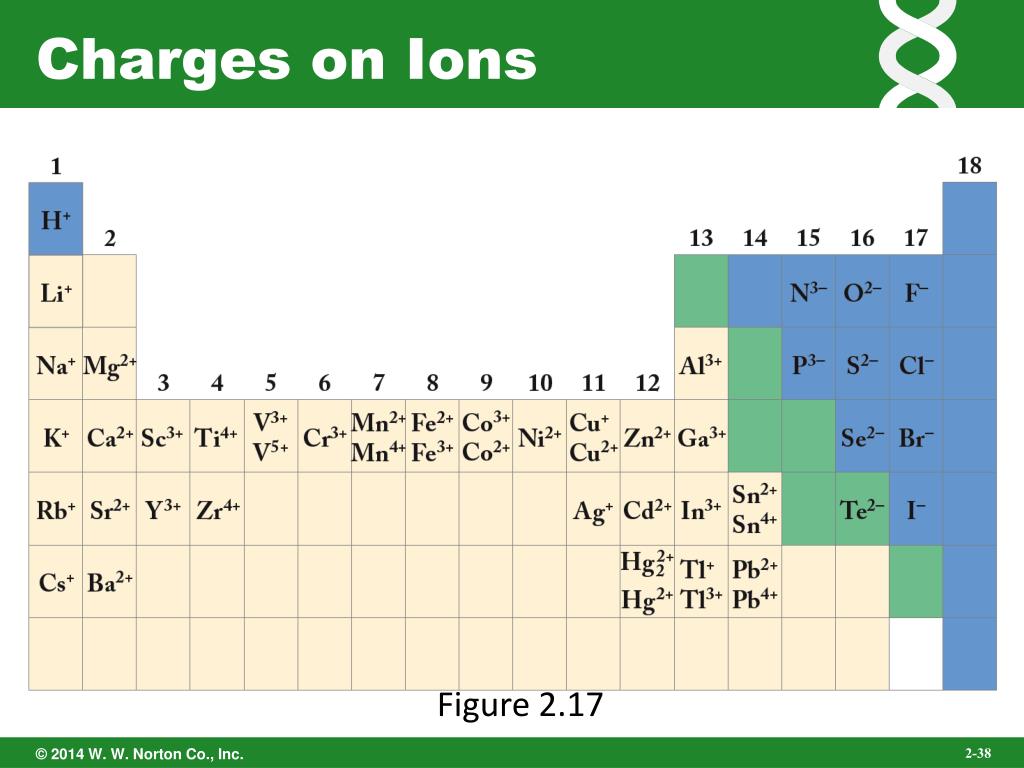
Periodic table with charged ions sciencetolf
The charge of an aluminum ion is determined by its electron. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
Protons, Neutrons, Electrons for Aluminum (Al, Al3+)
When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
What is the Charge on the Ion Formed by Aluminum? Aluminum Profile Blog
The charge of an aluminum ion is determined by its electron. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
How To Identify Charges Of Ions
Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). The charge of an aluminum ion is determined by its electron. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge.
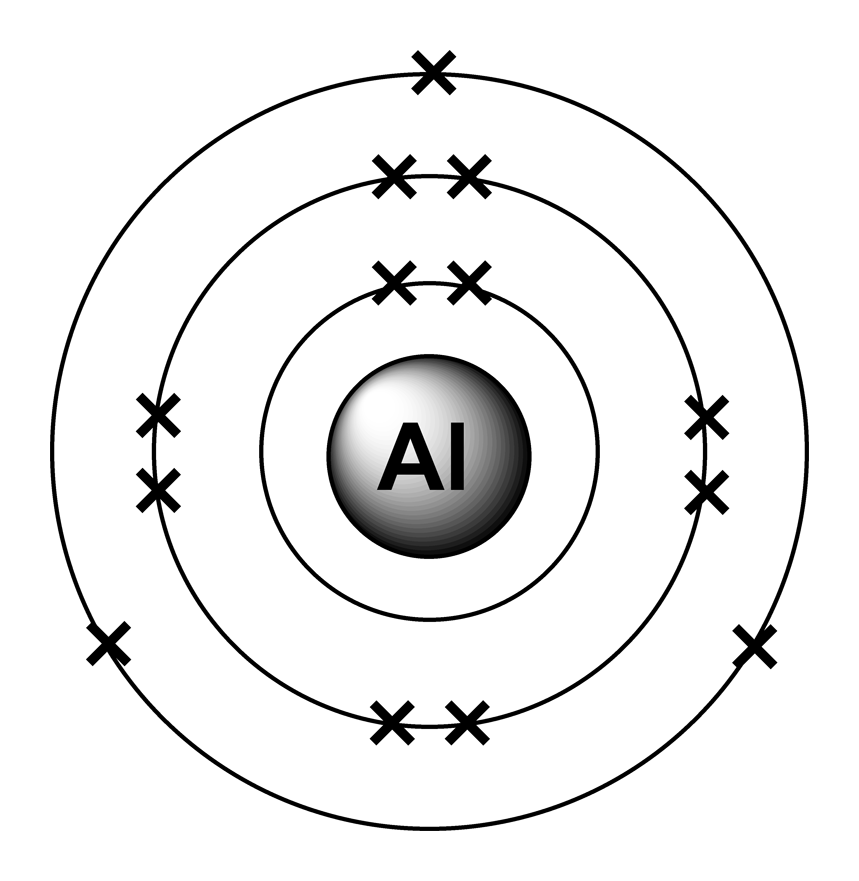
Electron arrangements
The charge of an aluminum ion is determined by its electron. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
Ion Charge from Periodic Table NemoQuiz
Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). The charge of an aluminum ion is determined by its electron. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge.
What is the Charge on the Ion Formed by Aluminum? Aluminum Profile Blog
When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).
Exploring Aluminum Ion Charge Properties, Characteristics and
Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron.
What is the Charge on the Ion Formed by Aluminum? Aluminum Profile Blog
Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺). When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. The charge of an aluminum ion is determined by its electron.
The Charge Of An Aluminum Ion Is Determined By Its Electron.
When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Aluminum ion loses 3 electrons, resulting in a charge of +3 (al³⁺).