What Does The Number Next To Isotopes Signify
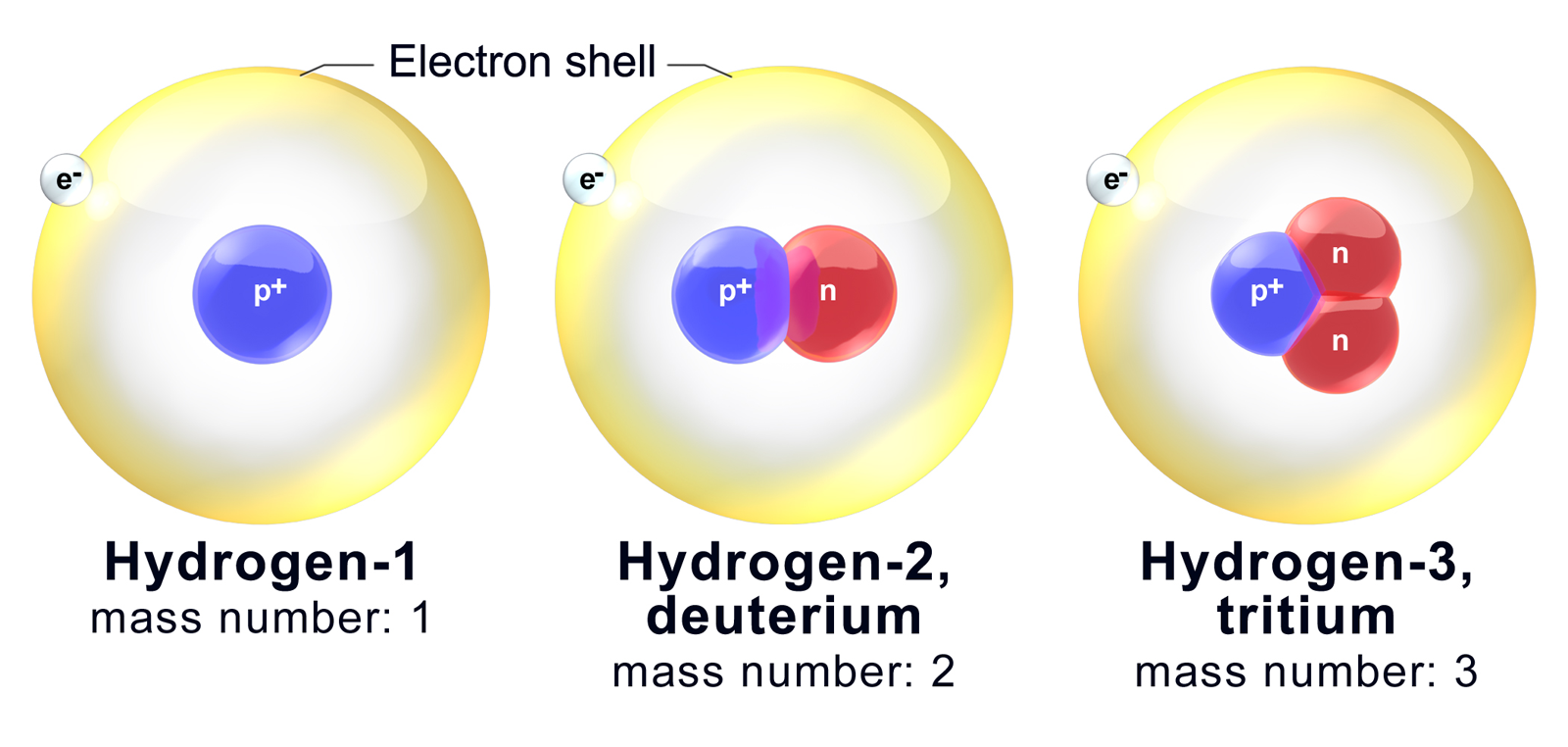
What Does The Number Next To Isotopes Signify - Isotopes are versions of the same element. Isotopes of an element have the same. The number next to isotopes signifies the mass number of the isotope. What does the number next to isotopes signify? Become a member and unlock all study. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. What does the number next to isotopes signify?(eg. It is called the mass number. The number indicates the isotope's mass number.
Isotopes of an element have the same. Become a member and unlock all study. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. The number next to isotopes signifies the mass number of the isotope. What does the number next to isotopes signify? Isotopes are versions of the same element. The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. It is called the mass number. What does the number next to isotopes signify?(eg. Isotopes are versions of the same element.
It is called the mass number. The number indicates the isotope's mass number. What does the number next to isotopes signify? The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. Isotopes are versions of the same element. The number next to isotopes signifies the mass number of the isotope. Isotopes are versions of the same element. Isotopes of an element have the same. Become a member and unlock all study. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the.
Practice Isotope Calculations 1 Worksheet Answers / Isotopes And Ions
Isotopes are versions of the same element. Isotopes of an element have the same. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. The number indicates the isotope's mass number. What does the number next to isotopes signify?(eg.
Solved 2. What does the number next to isotopes signify? For
The number next to isotopes signifies the mass number of the isotope. Isotopes are versions of the same element. What does the number next to isotopes signify? The number indicates the isotope's mass number. Isotopes are versions of the same element.
DOE Explains...Isotopes Department of Energy
Isotopes are versions of the same element. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. Become a member and unlock all study. What does the number next to isotopes signify?(eg. Isotopes are versions of the same element.
Isotopes Practice Worksheet Ions And Isotopes Practice Two Versions
What does the number next to isotopes signify?(eg. What does the number next to isotopes signify? Become a member and unlock all study. Isotopes are versions of the same element. The number next to isotopes signifies the mass number of the isotope.
PPT Isotopes PowerPoint Presentation, free download ID2481748
Isotopes are versions of the same element. What does the number next to isotopes signify?(eg. Isotopes are versions of the same element. The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. They have the same number of protons and electrons as the element but different mass numbers and number of.
SOLVED one of the isotopes of sulfur is represented as sulfur 35 or S
What does the number next to isotopes signify?(eg. What does the number next to isotopes signify? The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. Isotopes are versions of the same element. The number indicates the isotope's mass number.
Element Notation Examples
The number next to isotopes signifies the mass number of the isotope. The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. What does the number next to isotopes signify? It is called the mass number. Become a member and unlock all study.
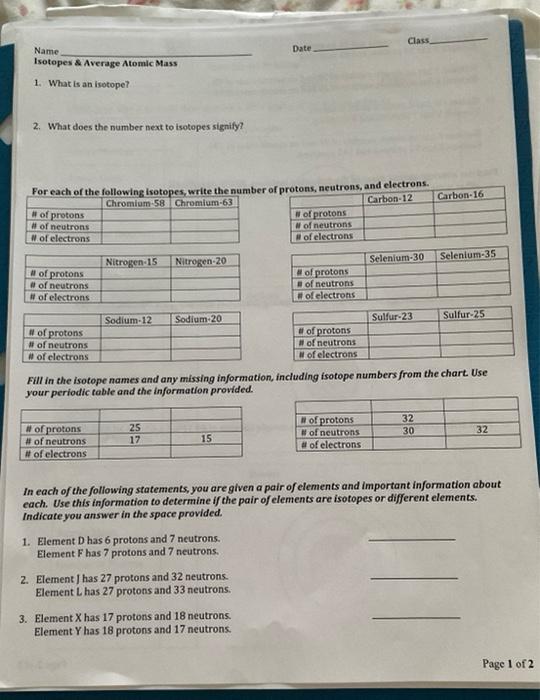
Answered 1. What is an isotope? 2. What does the… bartleby
The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. What does the number next to isotopes signify? Isotopes are versions of the same element. Become a member and.
Solved 2. What does the number next to isotopes signify? For
The number next to isotopes signifies the mass number of the isotope. Isotopes are versions of the same element. Isotopes of an element have the same. What does the number next to isotopes signify? The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom.
What is the isotope? Yoors
The number next to isotopes represents the sum of the protons and neutrons in the nucleus of the atom. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. What does the number next to isotopes signify? The number indicates the isotope's mass number. What does the number.
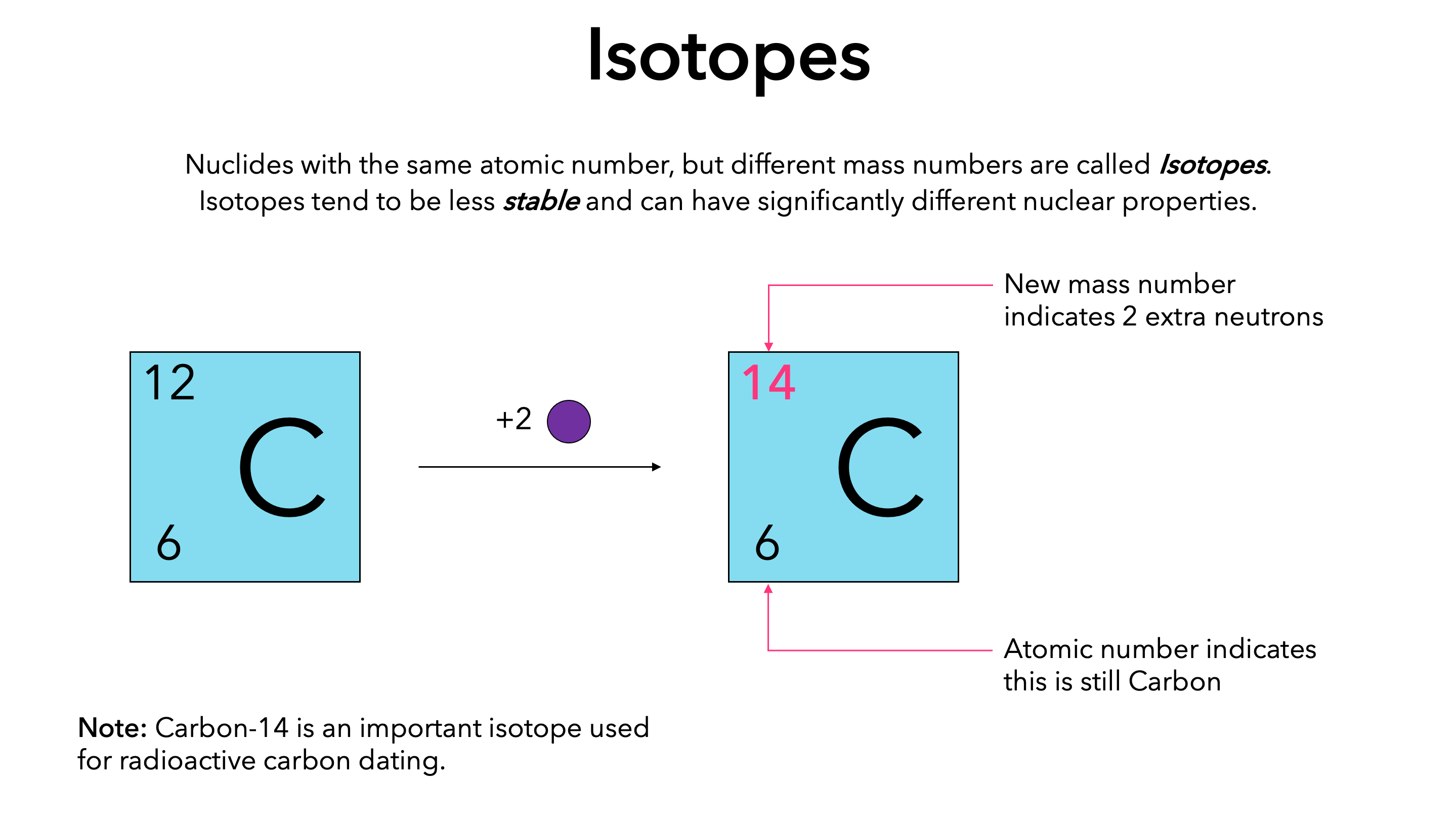
The Number Next To Isotopes Represents The Sum Of The Protons And Neutrons In The Nucleus Of The Atom.
What does the number next to isotopes signify?(eg. It is called the mass number. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.what does the. Isotopes of an element have the same.
The Number Indicates The Isotope's Mass Number.
The number next to isotopes signifies the mass number of the isotope. Isotopes are versions of the same element. Become a member and unlock all study. Isotopes are versions of the same element.