Paediatric Investigation Plan Template
Paediatric Investigation Plan Template - Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. For your submission, please use the latest versions of templates and forms, observing the drafting notes provided.
For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the.
• the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with.
Paediatric Investigation Plan Template
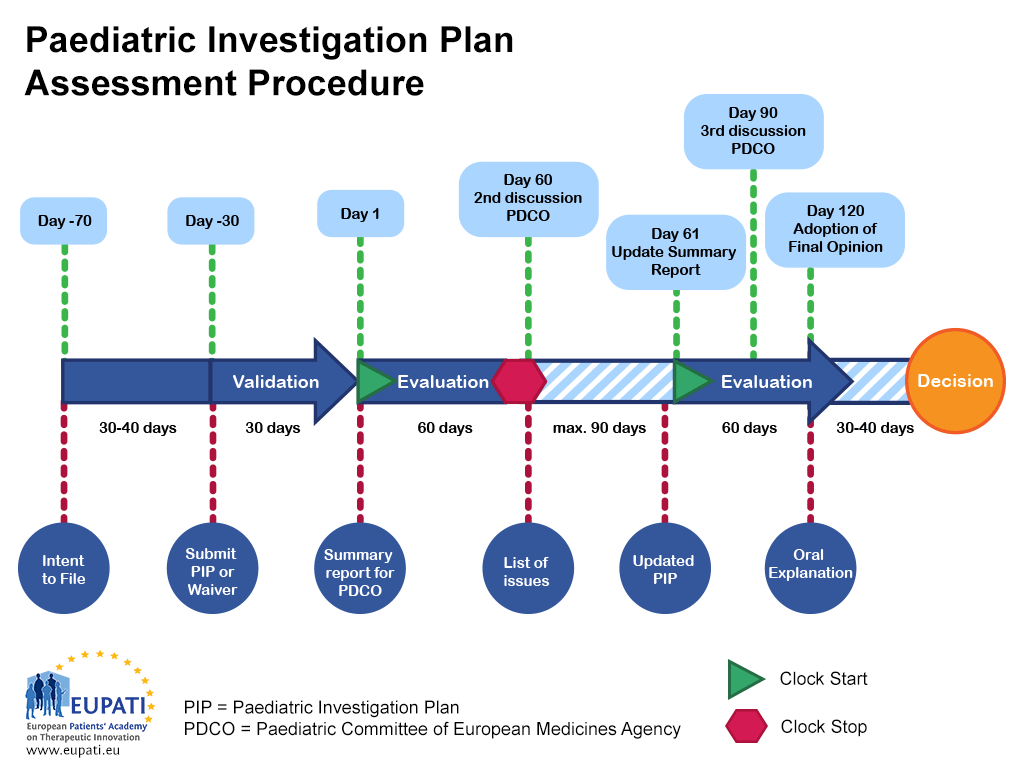
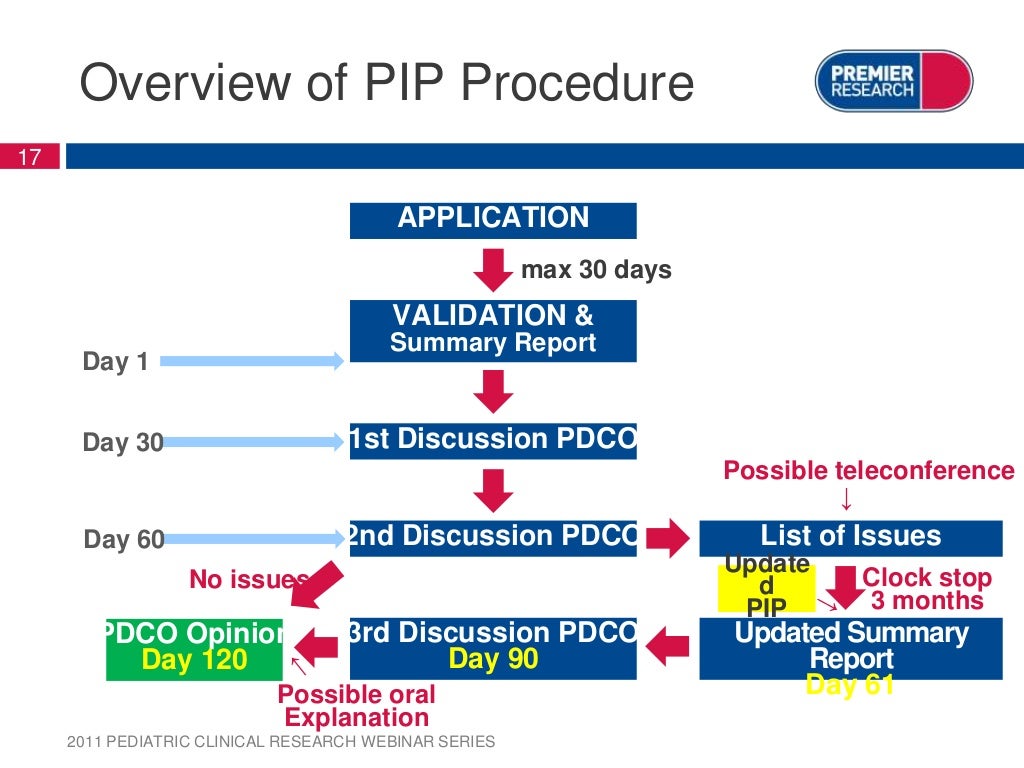
A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. A paediatric investigation plan (pip) is a development plan aimed at.
Paediatric medicine Paediatric Investigation Plan EUPATI
A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more.
Concept of Paediatric Investigation Plans conect4children
A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. A paediatric investigation plan is assessed by the paediatric committee of.
Paediatric Investigation Plan Template
A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more.
Paediatric Investigation Plan Template
For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary.
Paediatric Investigation Plan (PIP) Applications Steps to success
Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. •.
Paediatric Investigation Plan in Medicinal Product Development DDReg
A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. Pediatric.
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. • the development of paediatric regulations in the us over time • the definition, intent and timing of.
Concepts of Paediatric Investigation Plans (PIP) ppt download
• the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the. A paediatric investigation plan (pip) is a development plan aimed at ensuring that the necessary data are obtained through studies in. For your submission, please use the latest versions of templates and.
Paediatric Investigation Plan (PIP) Applications Steps to success
For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda,.
A Paediatric Investigation Plan (Pip) Is A Development Plan Aimed At Ensuring That The Necessary Data Are Obtained Through Studies In.
A paediatric investigation plan is assessed by the paediatric committee of the european medicines agency and follows a set procedure with. For your submission, please use the latest versions of templates and forms, observing the drafting notes provided. Pediatric studies under prea and potential pediatric uses under the bpca, is intended to result in a more efficient pediatric drug. • the development of paediatric regulations in the us over time • the definition, intent and timing of submission to the fda, and types of psps • the.