Gibbs Free Energy Of Formation Table
Gibbs Free Energy Of Formation Table - 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its.
100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its.
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat.
Standard Gibbs Free Energy Of Formation Table slideshare
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute.
Inspirational Standard Gibbs Free Energy Of formation Chart Check more
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g=.
Gibbs energy of formation values for the compounds involved in the
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g=.
Standard Gibbs Free Energy Of Formation Table slideshare
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute.
[PDF] Gibbs Free Energy of Formation for Selected Platinum Group
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of.
Standard Gibbs free energy of formation, standard enthalpy of formation
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of.
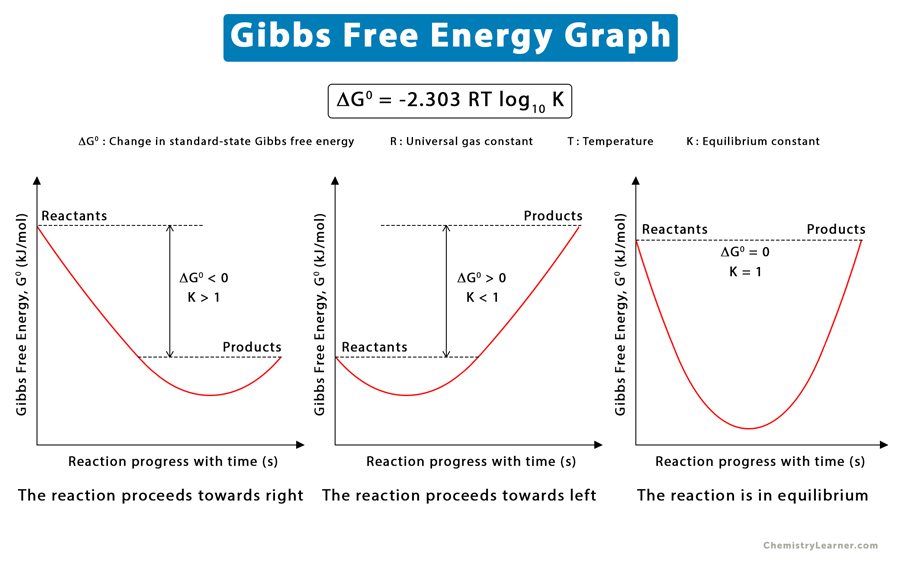
Gibbs Free Energy Definition, Equation, Unit, and Example
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g=.
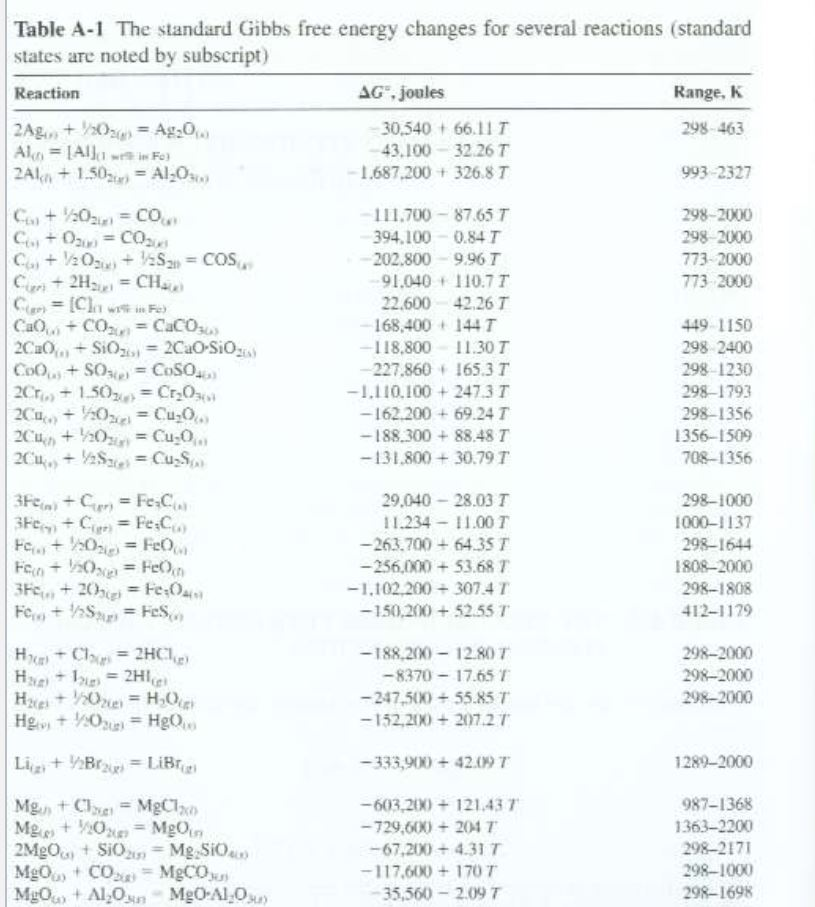
Table A1 The standard Gibbs free energy changes for
The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its. The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute.
Standard Gibbs Free Energy Of Formation Table slideshare
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g=.
Understanding Gibbs Free Energy Surfguppy Chemistry made easy for
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g=.
The Table Below Shows The Standard Enthalpy Of Formation, The Standard Gibbs Free Energy Of Formation, Standard Entropy And Molar Heat.
100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The standard gibbs free energy of formation at 25°c (298,15 k) for 1 mol of the substance in its given state (g= gas and l= liquide) from its.




![[PDF] Gibbs Free Energy of Formation for Selected Platinum Group](https://d3i71xaburhd42.cloudfront.net/1dbf18616943ea1a81a4ada05098657bbd1510ba/2-Table1-1.png)