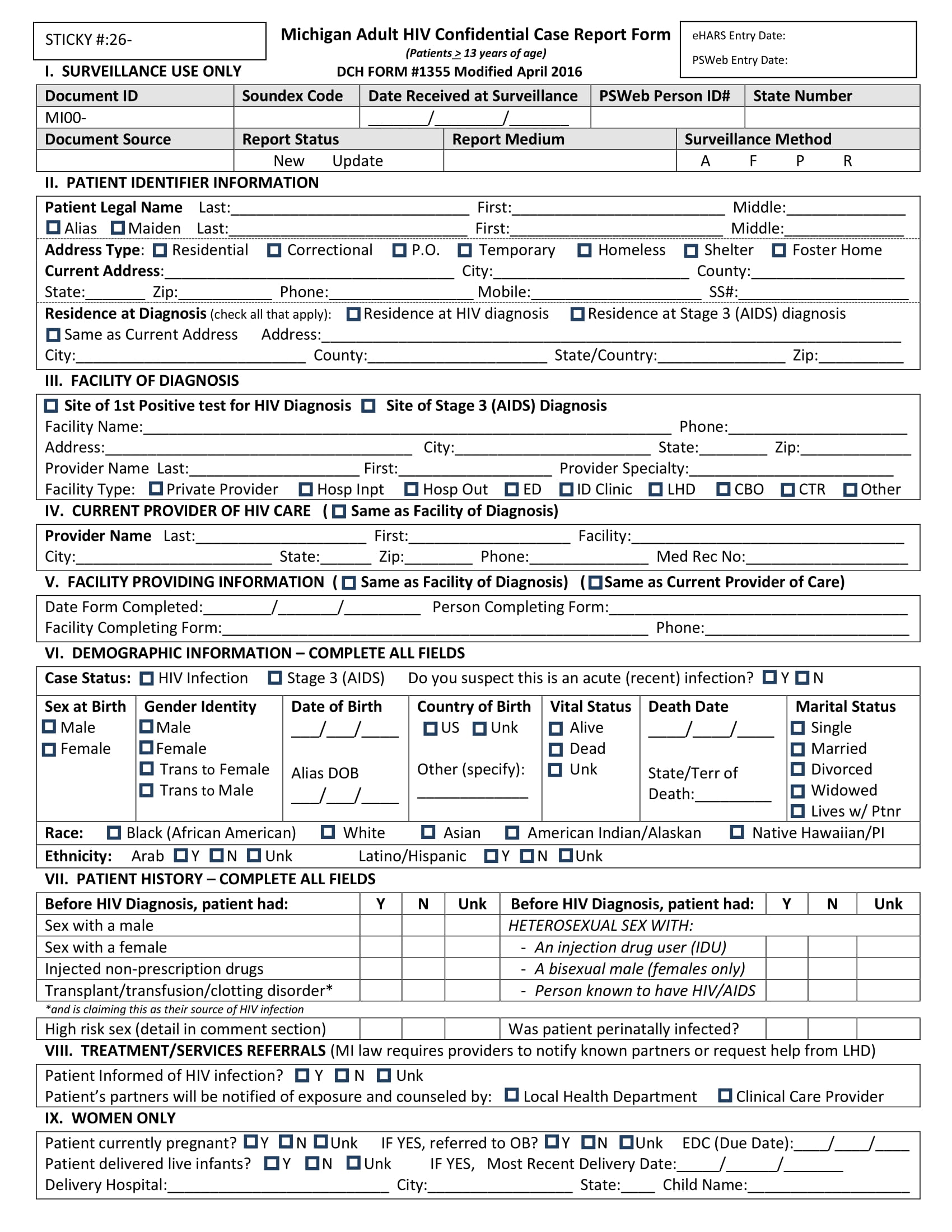
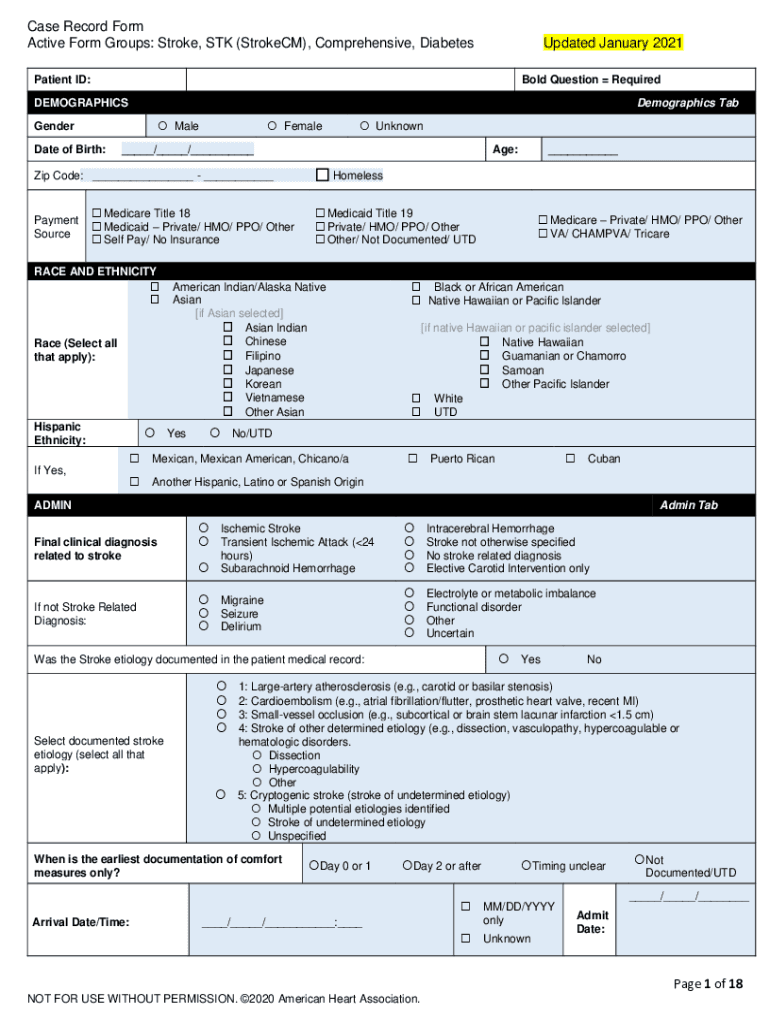
Case Record Form
Case Record Form - Find out the minimum information to. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. This presentation covers the key elements,. Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Find out the advantages and disadvantages of paper. Careful design of crfs is crucial for. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials.
This presentation covers the key elements,. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Find out the advantages and disadvantages of paper. Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Careful design of crfs is crucial for. Find out the minimum information to.
Case report forms (crfs) are critical documents in clinical research that record study data for each participant. This presentation covers the key elements,. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Careful design of crfs is crucial for. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Find out the minimum information to. Find out the advantages and disadvantages of paper.
Case Report Form ≡ Fill Out Printable PDF Forms Online
Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Careful design of crfs is crucial for. Learn what a case report form (crf) is, how to complete it, and why it is important.
FREE 15+ Case Report Forms in PDF MS Word
Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Careful design of crfs is crucial for. This presentation covers the key elements,. Find out the minimum information to. Learn how to design and develop a reliable and valid case report form (crf) for clinical research.
(PDF) CASE RECORD FORM INSTRUCTIONS SEVERE ACUTE … · This CRF is
Find out the advantages and disadvantages of paper. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Learn how to design and develop a reliable and valid case report form (crf) for clinical research. This presentation covers the key elements,. Case report forms (crfs) are critical documents in clinical.
FREE 15+ Case Report Forms in PDF MS Word
This presentation covers the key elements,. Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. Find out the minimum information to. Careful design of crfs is crucial for.
FREE 15+ Case Report Forms in PDF MS Word
Careful design of crfs is crucial for. This presentation covers the key elements,. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. Find out the minimum information to. Find out the advantages and disadvantages of paper.
CASE History b.ed CASE RECORD SECTIONI IDENTIFICATION DATA (CASE
Careful design of crfs is crucial for. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. Learn what a case record form (crf) is, how to design it, and what elements to include in it. This presentation covers the key elements,. Find out the minimum information to.
Printable Botox Treatment Record Template Printable Word Searches
Careful design of crfs is crucial for. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Case report forms (crfs) are critical documents in clinical research that record study data for each participant. This presentation covers the key elements,. Learn what a case report form (crf) is, how to complete.
Fillable Online Case Record Form Active Form Groups Stroke, Diabetes
Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. This presentation covers the key elements,. Careful design of crfs is crucial for. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Find out the advantages and disadvantages of paper.
Two pages of the case record form as an example. Download Scientific
Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Careful design of crfs is crucial for. Case report forms (crfs) are critical documents in clinical research that record study data.
Case Record Form
Case report forms (crfs) are critical documents in clinical research that record study data for each participant. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials. Careful design of crfs is crucial for. Learn what a case record form (crf) is, how to design it, and what elements to.
Careful Design Of Crfs Is Crucial For.
Find out the minimum information to. This presentation covers the key elements,. Learn what a case record form (crf) is, how to design it, and what elements to include in it. Case report forms (crfs) are critical documents in clinical research that record study data for each participant.
Find Out The Advantages And Disadvantages Of Paper.
Learn how to design and develop a reliable and valid case report form (crf) for clinical research. Learn what a case report form (crf) is, how to complete it, and why it is important for clinical trials.