Can P Orbitals Form Sigma Bonds
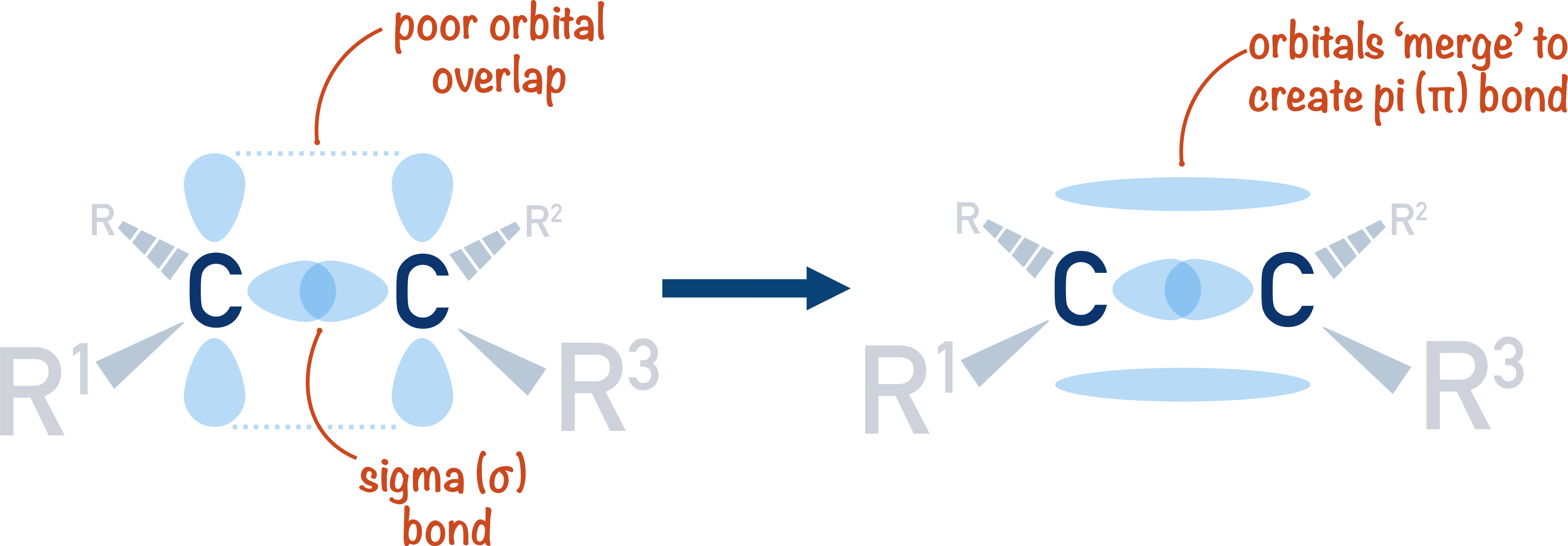
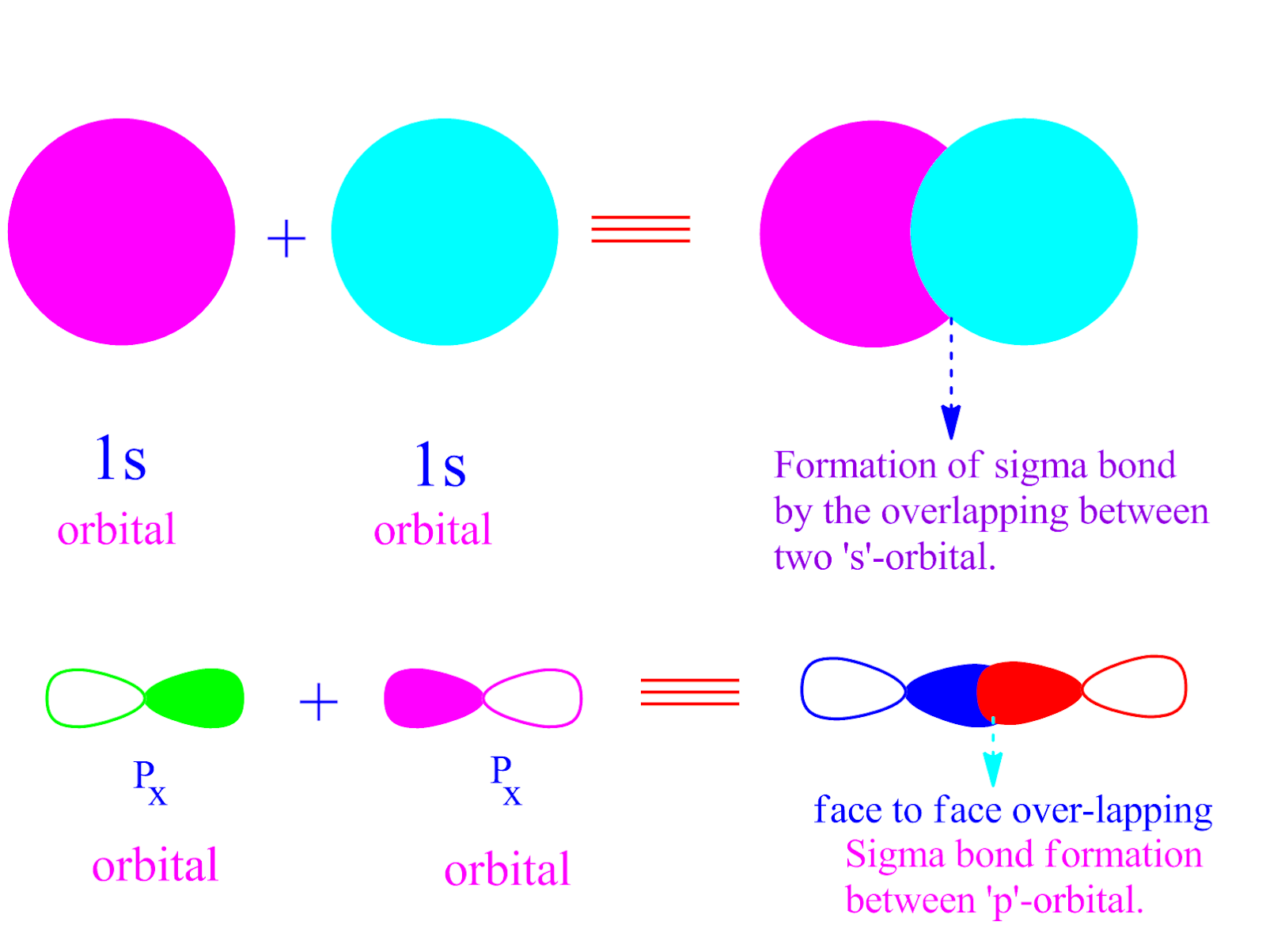
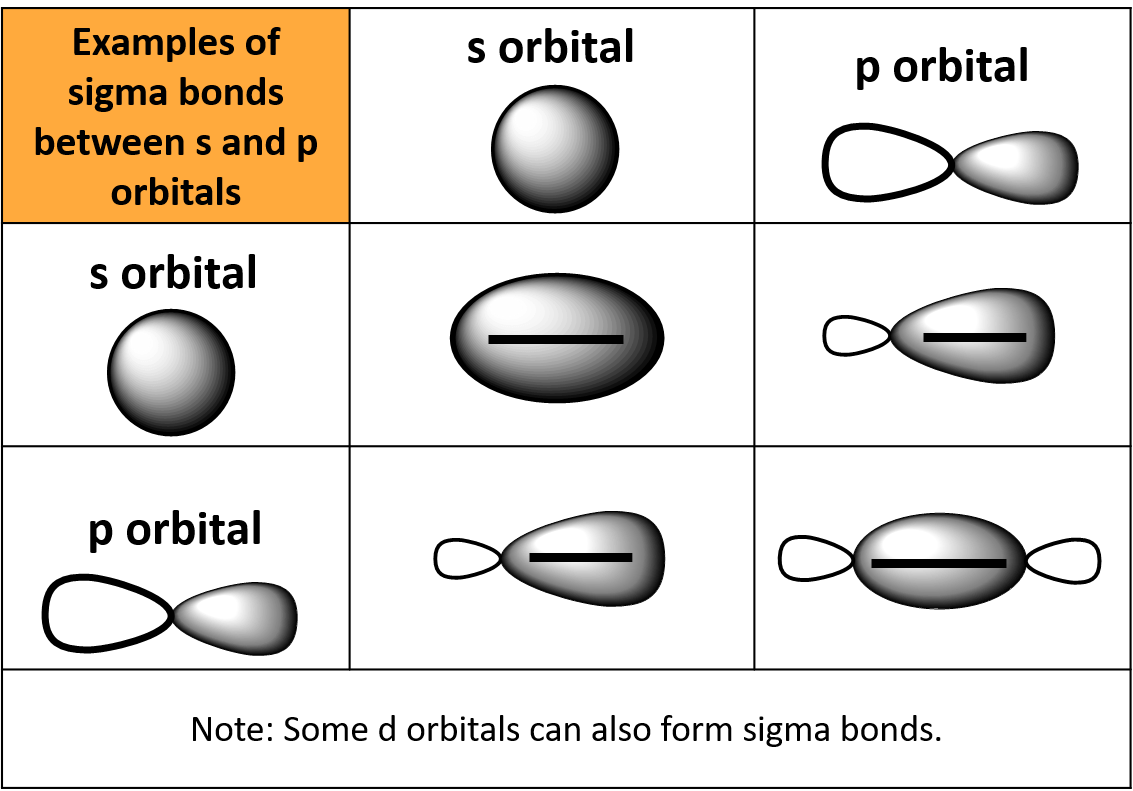
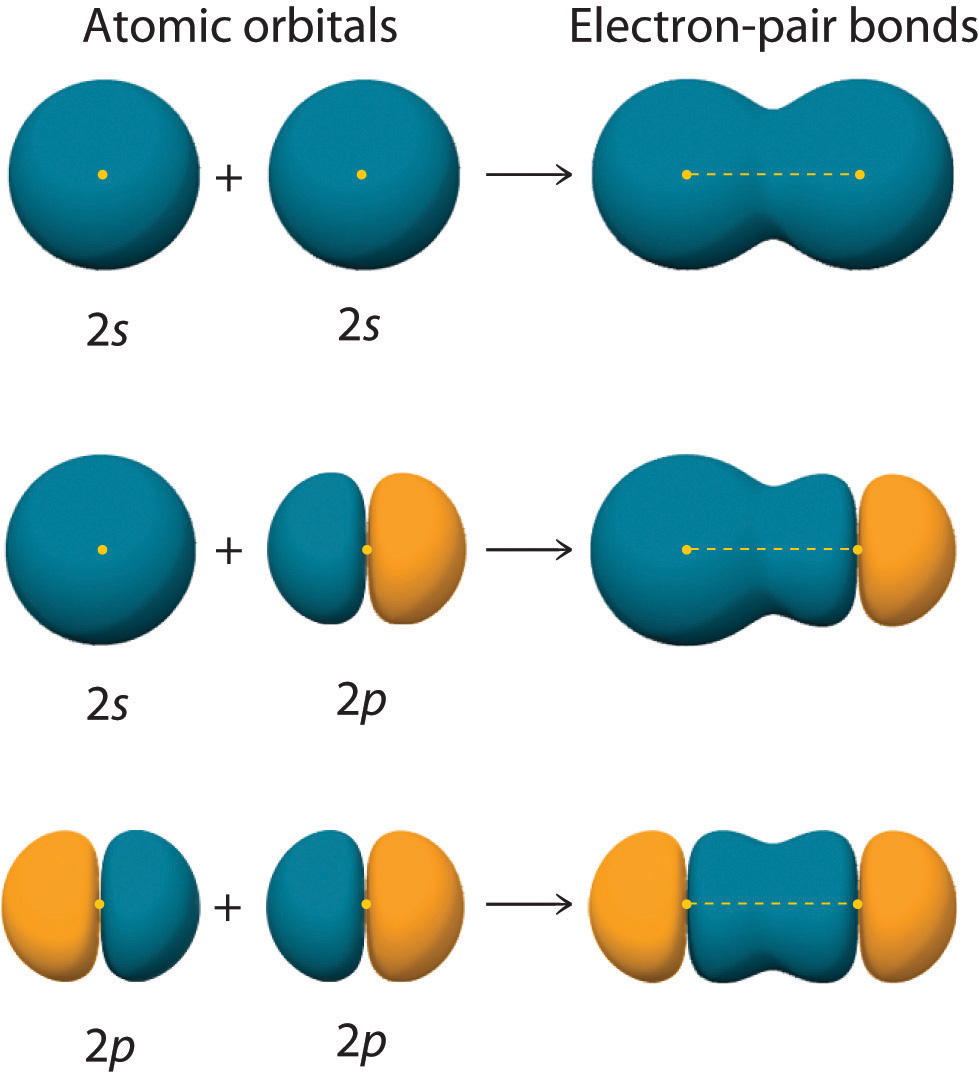
Can P Orbitals Form Sigma Bonds - This type of overlap allows the electron density to be concentrated along. A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. A sigma bond can also be formed by the overlap of two p orbitals.
This type of overlap allows the electron density to be concentrated along. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. A sigma bond can also be formed by the overlap of two p orbitals. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. A pi (π) orbital is one that has one node.
Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. This type of overlap allows the electron density to be concentrated along. A pi (π) orbital is one that has one node. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. A sigma bond can also be formed by the overlap of two p orbitals.
Alkenes (ALevel) ChemistryStudent
A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. This type of overlap allows the electron density to be concentrated along. A sigma bond can also be formed by the overlap of two p orbitals. The covalent bond in molecular.
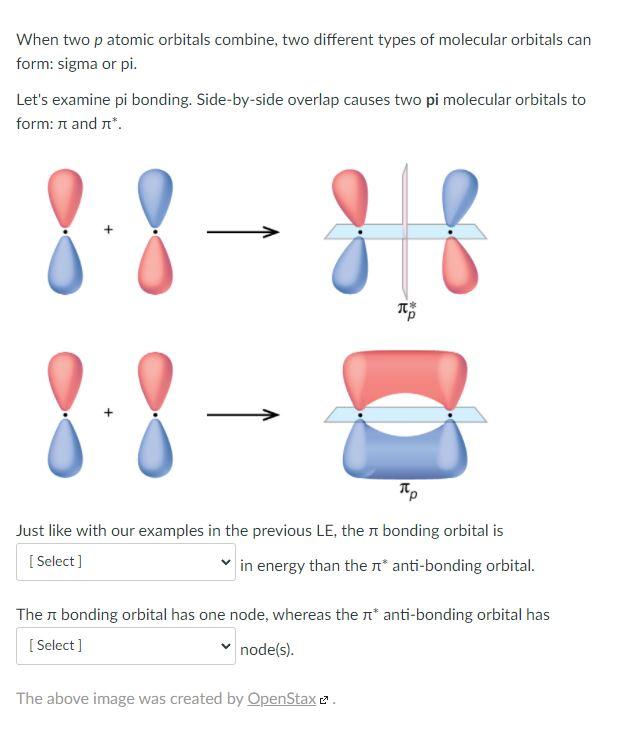
A π bond is formed by the overlap of
The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. This type of overlap allows the electron density to be concentrated along. A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. A sigma bond can.
Why are sigma bond more stronger than pi bond ? PG.CHEMEASY
The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. A pi (π) orbital is one that has one node. This type of overlap allows the electron density to be concentrated along. A sigma bond can.
Solved When two p atomic orbitals combine, two different
This type of overlap allows the electron density to be concentrated along. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. A sigma bond can.
Sigma和π键聪明的数学和科学Wiki
Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. This type of overlap allows the electron density to be concentrated along. A sigma bond can also be formed by the overlap of two p orbitals. A pi (π) orbital is one that has one node. The covalent bond in molecular.
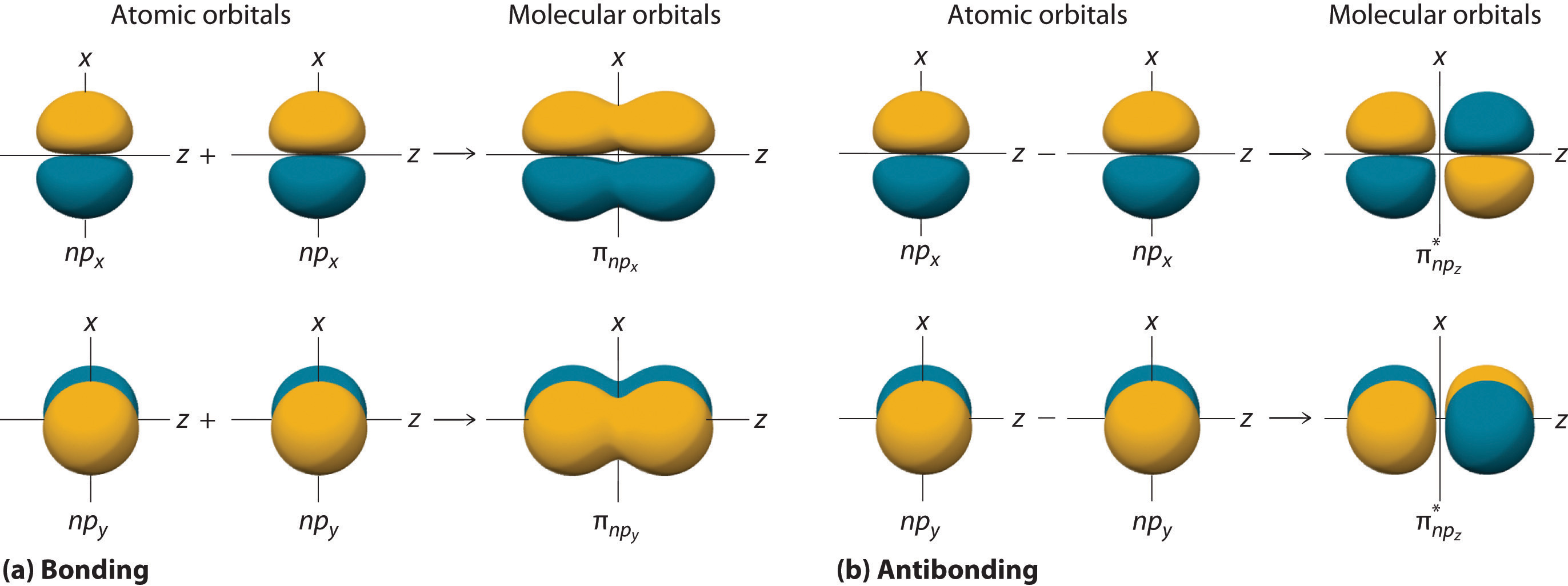
9.3 Molecular Orbital Theory Chemistry LibreTexts
The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. This type of overlap allows the electron density to be concentrated along. A sigma bond can also be formed by the overlap of two p orbitals. A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are.
8 Drawing Molecular Orbital Diagrams — Flux Science
A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. A sigma bond can also be formed by the overlap of two p orbitals. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. This type.
Pi Bond And Sigma Bond How to count sigma and pi bonds Quora 3
A pi (π) orbital is one that has one node. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. This type of overlap allows the electron density to be concentrated along. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. A sigma bond can.
2p Orbitals
This type of overlap allows the electron density to be concentrated along. A sigma bond can also be formed by the overlap of two p orbitals. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the..
sigma /r/okbuddyretard OkBuddyRetard Know Your Meme
A sigma bond can also be formed by the overlap of two p orbitals. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. This type of overlap allows the electron density to be concentrated along..
This Type Of Overlap Allows The Electron Density To Be Concentrated Along.
A pi (π) orbital is one that has one node. A sigma bond can also be formed by the overlap of two p orbitals. The covalent bond in molecular fluorine, f 2, is a sigma bond formed by the. Any two orbitals (s, p, or d) that are oriented along the bonding axis can form a sigma bond.