Can Acetone Form Hydrogen Bonds
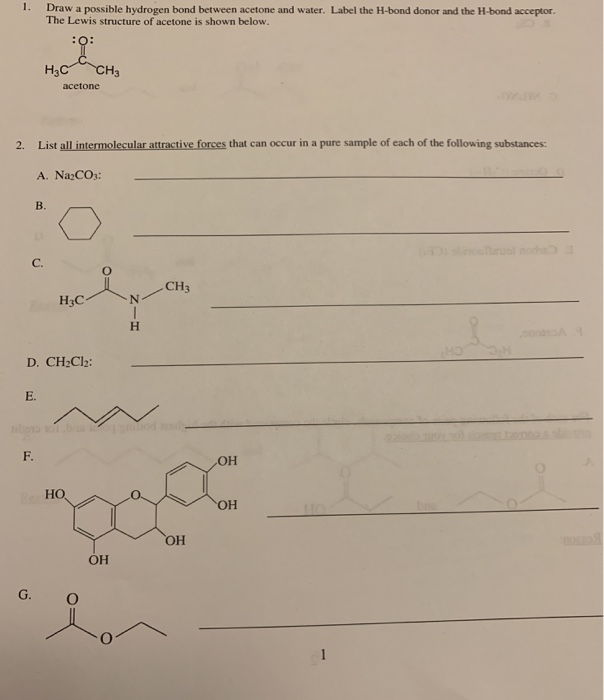
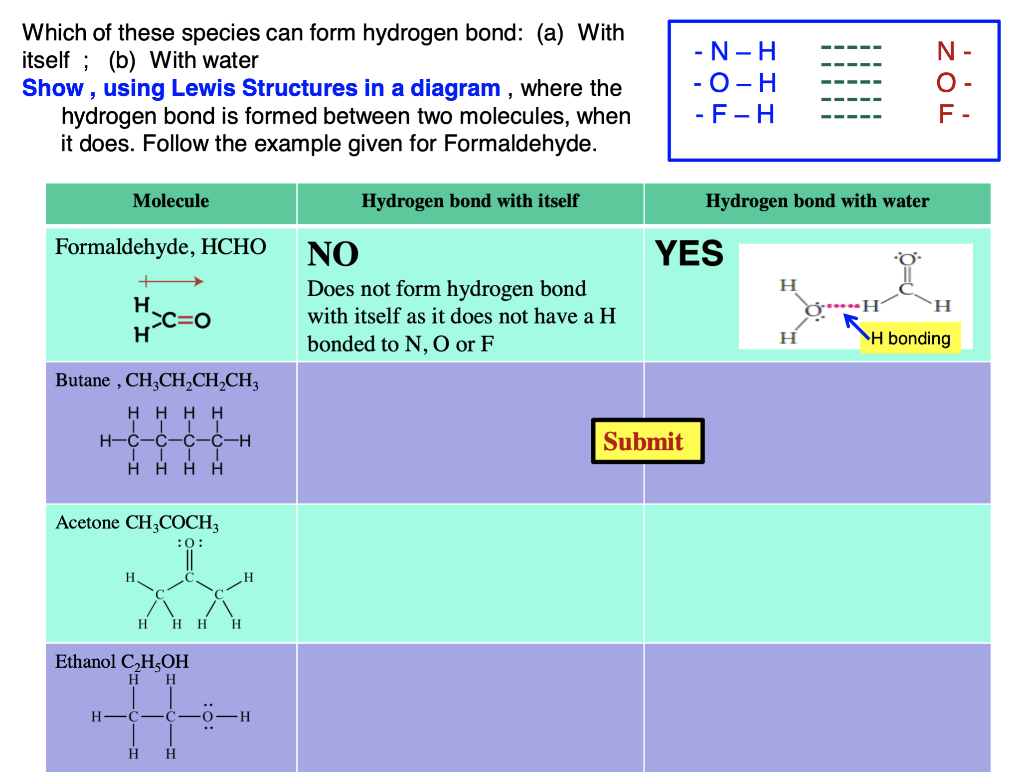
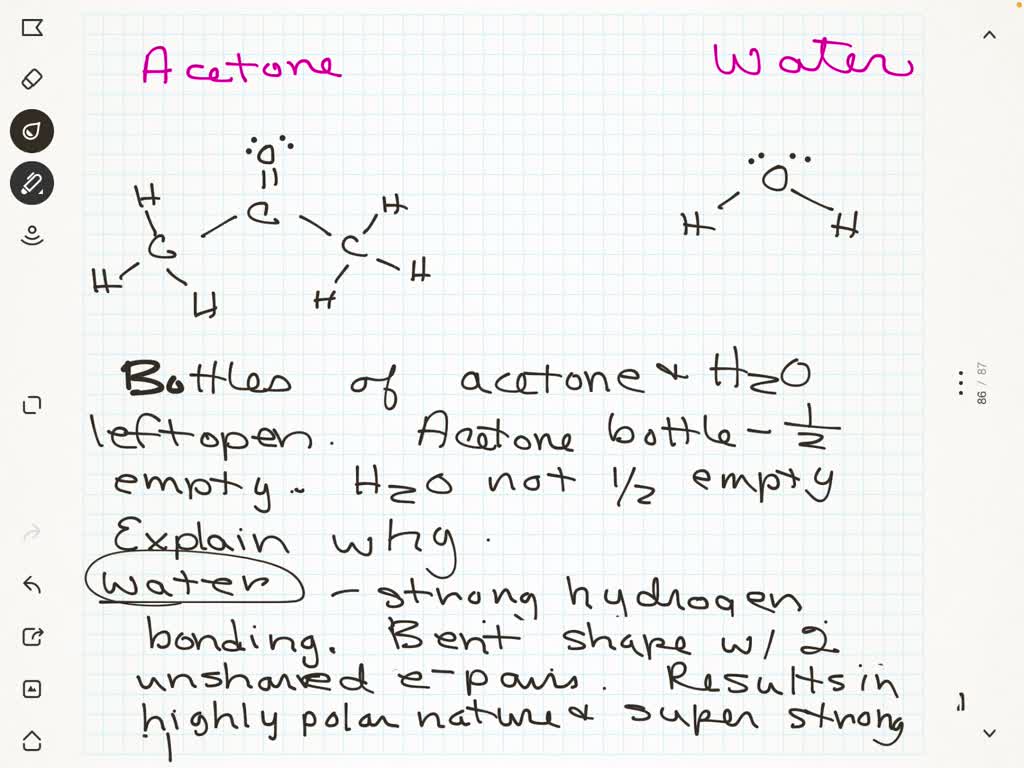
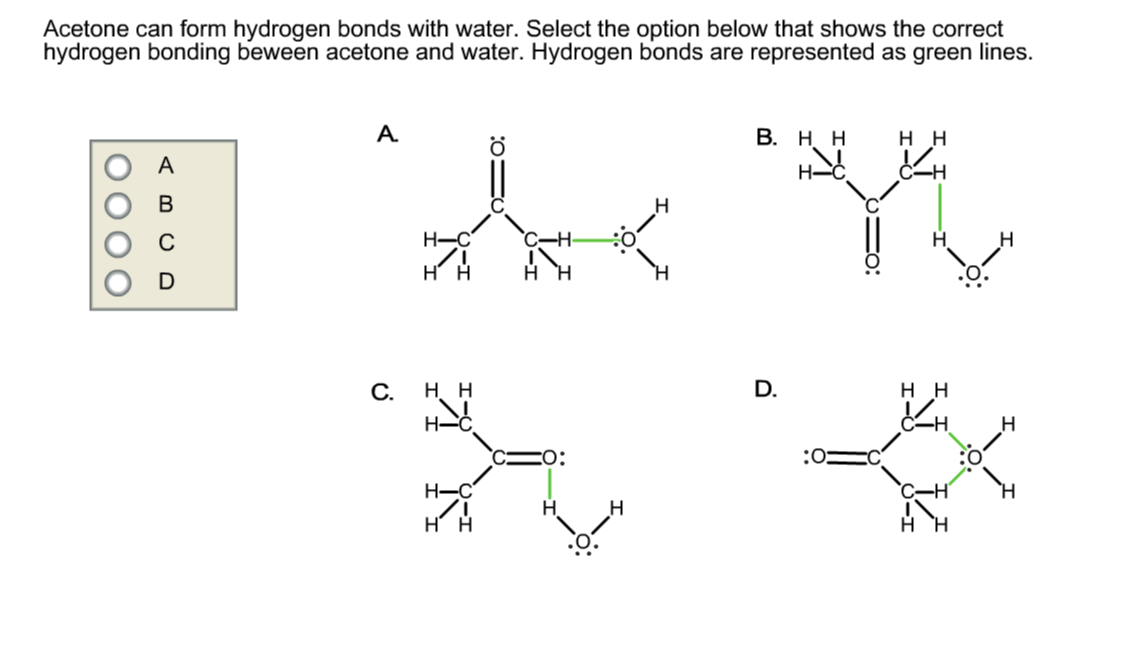
Can Acetone Form Hydrogen Bonds - Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. Acetone can form hydrogen bonds with water. The water molecule forms two hydrogen bonds with acetone: Four images are given which show an acetone molecule interacting with a water. I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,.
The water molecule forms two hydrogen bonds with acetone: The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Four images are given which show an acetone molecule interacting with a water. Acetone can form hydrogen bonds with water. I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming.
Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. Acetone can form hydrogen bonds with water. The water molecule forms two hydrogen bonds with acetone: I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. Four images are given which show an acetone molecule interacting with a water. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,.
Acetone Lewis Structure With Polarity
The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Four images are given which show an acetone molecule interacting with a water. Acetone can form hydrogen bonds with water. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. The water molecule forms two.
Solved 1. Draw a possible hydrogen bond between acetone and
The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Four images are given which show an acetone molecule interacting with a water. Acetone can form hydrogen bonds with water. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. I haven't been able to.
SOLVED How many hydrogen bonds can form between an acetone molecule
The water molecule forms two hydrogen bonds with acetone: Acetone can form hydrogen bonds with water. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. Four images are given which show an acetone molecule interacting with a water. The hydrogen atom of h 2 o is coordinated to the.
Answered Can Butane, Acetone, and Ethanol form a… bartleby
Acetone can form hydrogen bonds with water. The water molecule forms two hydrogen bonds with acetone: Four images are given which show an acetone molecule interacting with a water. I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,.
SOLVED2. Can a molecule of acetone form a hydrogen bond with water
Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. Acetone can form hydrogen bonds with water. Four images are given which show an acetone molecule interacting with a water. The water molecule forms two hydrogen bonds with acetone: I haven't been able to find a reference confirming that fluoroform.
SOLVED SOURCE CHAPTER PROBLEM TOPIC INTRAMOLECULAR FORCES The
The water molecule forms two hydrogen bonds with acetone: The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. Acetone can form hydrogen bonds with water. Four images are given which show an acetone molecule interacting with a water.
Solved Acetone can form hydrogen bonds with water. Select
Acetone can form hydrogen bonds with water. I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. The water molecule forms two hydrogen bonds with acetone: Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. Four images are given which show an acetone.
hydrogen bond acetone ethanol YouTube
The water molecule forms two hydrogen bonds with acetone: I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,..
SOLVEDWhy can't two molecules of acetone form a hydrogen bond with
Four images are given which show an acetone molecule interacting with a water. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Acetone can form hydrogen bonds with water. The water molecule forms two hydrogen bonds with acetone: I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone.
I had to draw Hbonds between water and Acetone. Did I do it correctly
I haven't been able to find a reference confirming that fluoroform forms hydrogen bonds with acetone. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Four images are given which show an acetone molecule.
The Water Molecule Forms Two Hydrogen Bonds With Acetone:
Four images are given which show an acetone molecule interacting with a water. Acetone can form hydrogen bonds with water. The hydrogen atom of h 2 o is coordinated to the carbonyl group of acetone,. Acetone does not have a hydrogen atom bonded to a highly electronegative atom, which is the primary requirement for forming.