Add Formal Charges To Each Resonance Form Of Nco
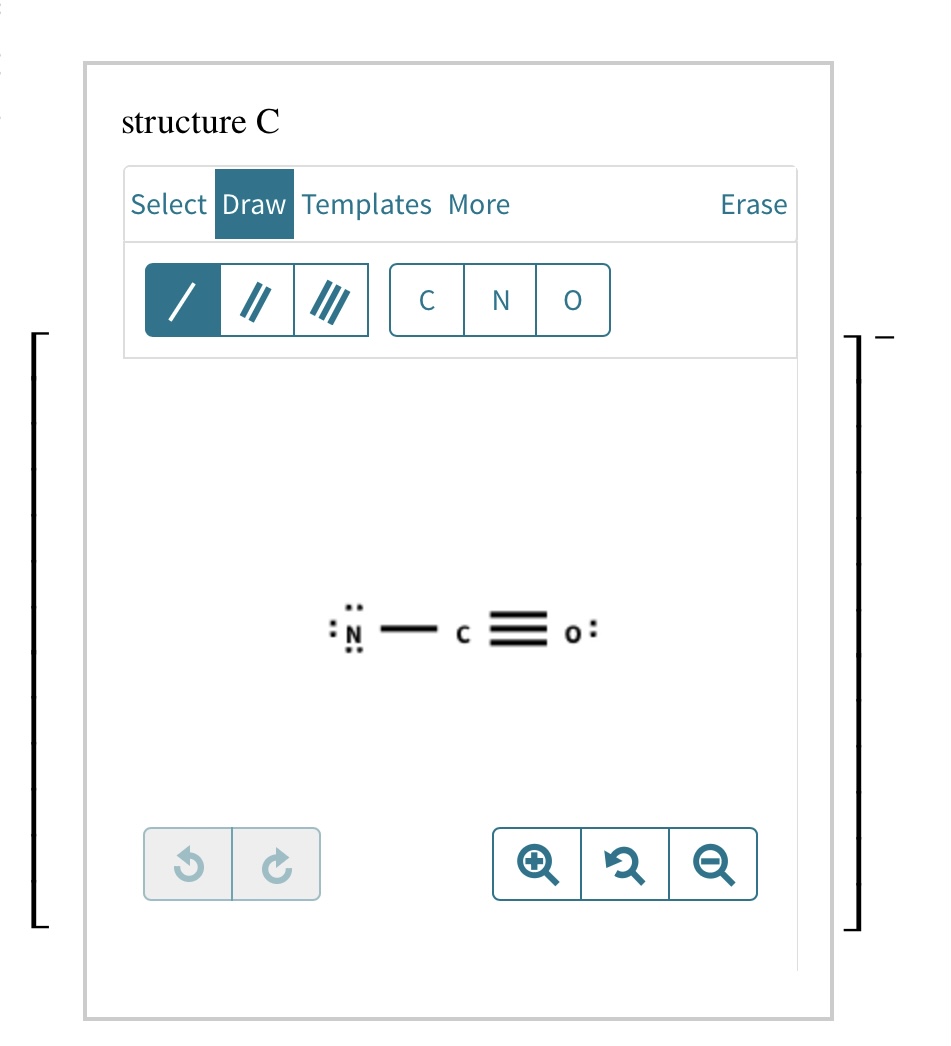
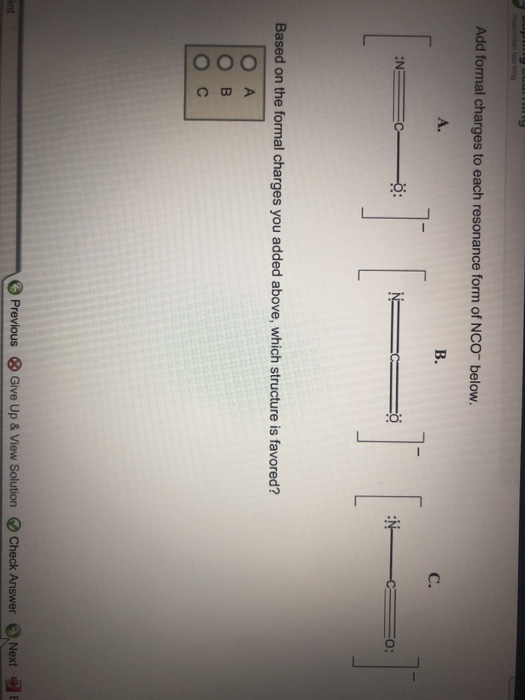
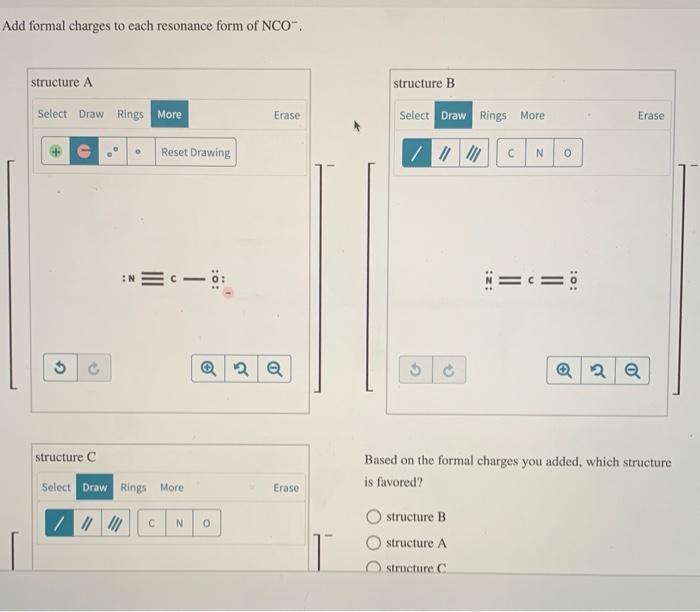
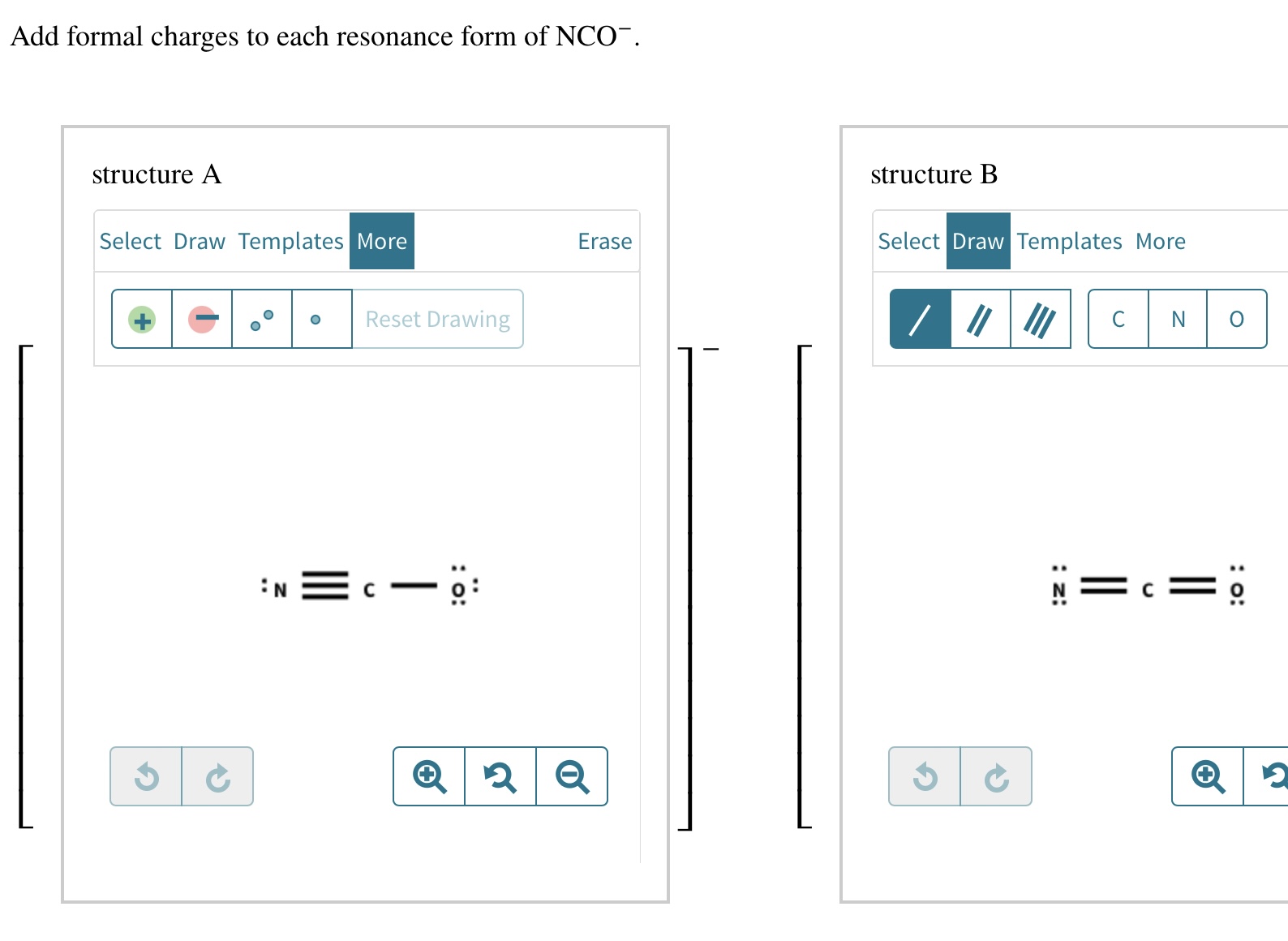
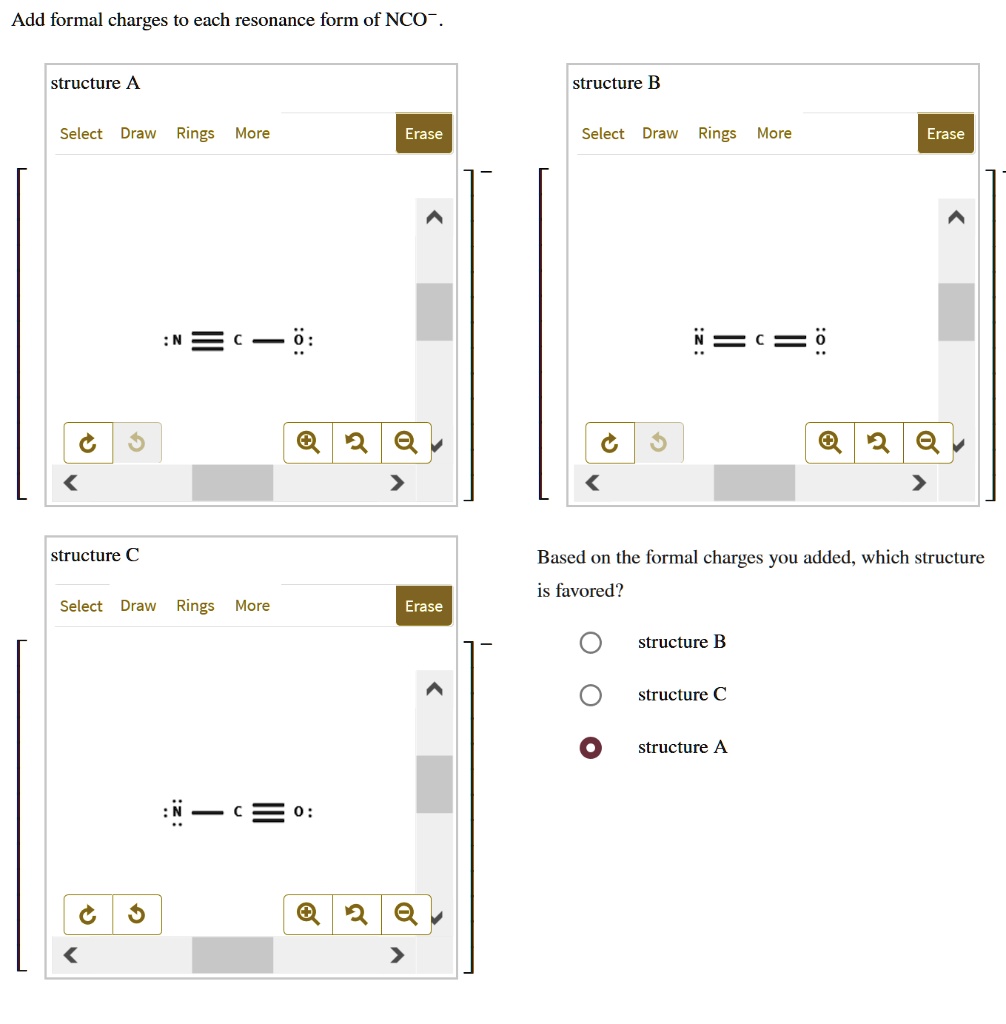
Add Formal Charges To Each Resonance Form Of Nco - Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th.
Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
[Solved] Add formal charges to each resonance form of NCO structure A
Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
Solved Add formal charges to each resonance form of
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th.
Solved y Add formal charges to each resonance form of NCO
Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
add formal charges to each resonance form of HCNO below WizEdu
Given to add formal charge to each resonance form of nco a −. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Based on the formal charge need to determine th.
18 Captivating Facts About Formal Charge
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th.
Answered Add formal charges to each resonance… bartleby
Based on the formal charge need to determine th. Given to add formal charge to each resonance form of nco a −. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
Solved Add formal charges to each resonance form of NCO.
Based on the formal charge need to determine th. Given to add formal charge to each resonance form of nco a −. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero.
Solved Add formal charges to each resonance form of
Given to add formal charge to each resonance form of nco a −. When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Based on the formal charge need to determine th.
Chemistry Archive January 26, 2015
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Based on the formal charge need to determine th. Given to add formal charge to each resonance form of nco a −.
SOLVED Add formal charges to each resonance form of NCO structure A
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Given to add formal charge to each resonance form of nco a −. Based on the formal charge need to determine th.
Given To Add Formal Charge To Each Resonance Form Of Nco A −.
When determining the best lewis structure or the predominant resonance structure, we want that the atoms have a formal charge close to zero. Based on the formal charge need to determine th.